The delta32 mutation sounds really great. Can we give everyone this mutation?
May 10, 2005

- Related Topics:
- Genetic engineering,
- Futuristic science,
- Medical genetics,
- Bioethics
A high school teacher from California asks:
"What a great mutation the delta32 mutation sounds like. No side effects and it makes you resistant to HIV. Is there some way to give everyone this mutation? Is there anywhere to get tested for it?
Maybe somehow they could utilize early embryonic cells and transform them to incorporate all or part of the mutation in each sequential cell line from then on. Or how about using an engineered smallpox or similar virus as a vector to infect these embryonic cells?"
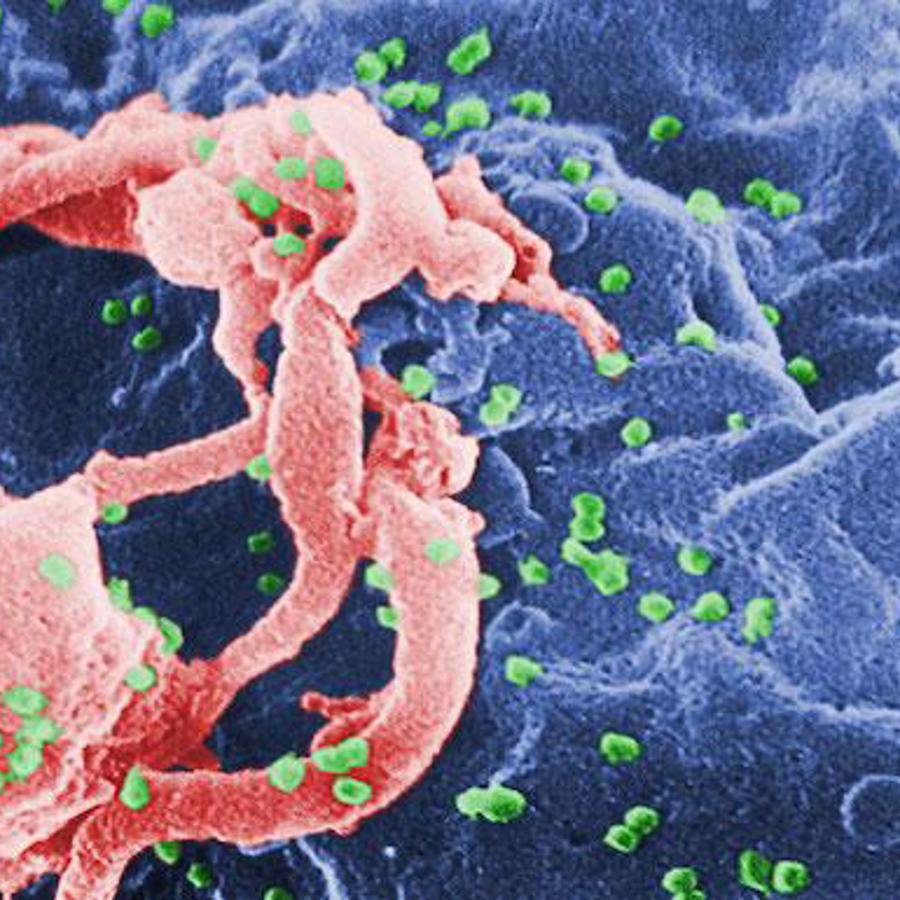
It does sound like a wonderful mutation doesn't it? The CCR5-delta32 mutation not only may make us resistant to HIV but maybe smallpox and the plague too (see the links below for more details).
So, can we get this thing into our DNA? Or at least our kids? Maybe in the future, but I don't see it happening anytime soon. Getting new DNA into a human safely and effectively just isn't yet a reality, and it is a controversial topic.
First off, current gene therapy tools aren't perfect. For starters, they insert DNA randomly into cells. What that means is that a new gene could land anywhere in the DNA.
If it lands in the wrong place, things can go wrong pretty quickly. One of the most common problems is that the body shuts the gene off. The body does this to foreign genes because it treats them like an invader.
Many viruses, like HIV, stick themselves into our DNA. Once this happens, they turn on their genes and crank out harmful proteins. It is usually in the body's best interest to shut off these sorts of genes.
Sometimes even worse things can happen. In one gene therapy experiment in France, the gene landed in the wrong spot and caused leukemia. How did it do this?
The virus carrying the gene landed near an oncogene. An oncogene is a gene that can cause cancer. By landing next to this oncogene, the virus turned it on and the kids got cancer.
OK, let's say we did get it into the cell and it stayed on and didn't have some awful side effect. Would it work then?
In this case, probably not. You most likely would end up with some copies of the normal CCR5 gene and some copies with the delta32 mutation. To get the full effect of the delta32 mutation, you need to eliminate any normal CCR5 that is around.
Having one copy of the delta32 mutation gives some protection against HIV. It also makes the disease less severe once you get it. But this probably isn't good enough to be worth the risks of gene therapy.
What about putting the delta32 deletion into our own copies of the CCR5 gene? Now we're getting into a tough area.
As you may know, scientists have been able to shut off genes in mice for many years now. The gene is "knocked out" by sticking a piece of DNA into the middle of the gene. It is really hard to do but worth it because we can learn so much.
It may be enough to knock out the CCR5 gene and not to recreate the delta32 mutation. This is because the delta32 mutation essentially wipes out the activity of the CCR5 gene.
OK, can we do it in people? Not ethically (and maybe not scientifically). With a mouse, the first step only creates a chimera. What this means is that the mouse has some cells that have the gene knocked out and some that don't.
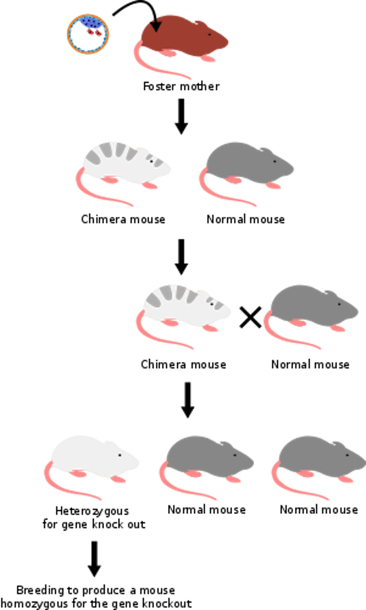
You then pick the mice that can pass the knocked out gene to its pups. Of course, only one of the genes is usually knocked out at this point. To get both knocked out, you then need to breed the new pups and pick those with both genes knocked out. Around 1 in 4 pups of this second generation should have both copies knocked out.
Obviously we can't do this with people. But it may be possible to do it just in a petri dish. Since the CCR5 protein is on blood cells, maybe we could just knock CCR5 out in the stem cells in bone marrow. You'd probably have to wipe out a person's existing bone marrow and replace it with the mutant version, though (which could have unintended consequences).
Another option is a bone marrow transplant from a donor who has two copies of the delta32 mutation. This has actually been done in at least one patient, and seems to have cured him of AIDS (he was being treated for leukemia, and the delta32 replacement was a fortunate side effect of the treatment).1 Of course, bone marrow transplants are a pretty big deal with a high mortality rate.
None of this sounds trivial does it? Gene editing can have significant consequences, and we don’t even fully understand what it could do in humans. It just doesn't seem like it is happening soon.
Another way to make sure a child inherits it is for both parents to each have at least one copy. If the parents each have two copies, then all of their kids will inherit the CCR5-delta32 mutation too.
If either or both parents only have one copy, then a test like preimplantation genetic diagnosis (PGD) could be used to pick the embryos with two copies of the delta32 version of the CCR5 gene.2 This is beginning to tread on ethically shaky ground, but it is scientifically possible.
So there you have it. Right now, the technology just isn't ready for even the brute force of knocking out a gene. There are some newer technologies like RNAi or chimeric DNA/RNA hybrids that might work in the future, but nothing right now. And even if these new methods work, the ethics and side effects of editing human genes need to be seriously considered.
Read More:
- Dr. Dan Stowell: The Molecules of HIV
- CCR5, the plague, and smallpox
- NIEHS: knocking out genes in mice
- Science: Jail time for scientist who created embryos with this change
- Kitty Cooper: Checking your HIV resistance with 23andMe

Author: Dr. Barry Starr
Barry served as The Tech Geneticist from 2002-2018. He founded Ask-a-Geneticist, answered thousands of questions submitted by people from all around the world, and oversaw and edited all articles published during his tenure. AAG is part of the Stanford at The Tech program, which brings Stanford scientists to The Tech to answer questions for this site, as well as to run science activities with visitors at The Tech Interactive in downtown San Jose.
 Skip Navigation
Skip Navigation
